Home|Blog | FDA Approves New Natrelle INSPIRA® Breast Implants
FDA Approves New Natrelle INSPIRA® Breast Implants
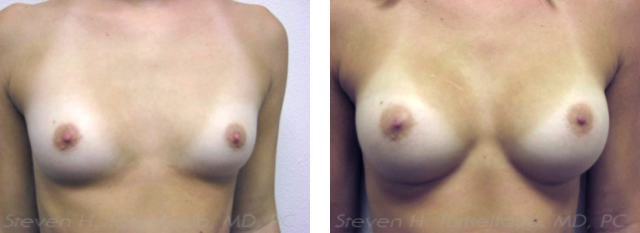
The U.S. Food and Drug Administration (FDA) recently approved a new line of silicone gel breast implants produced by Allergan that are marketed under the name Natrelle INSPIRA® Cohesive breast implants. These new implants offer women an even greater diversity of choices that can potentially provide better and more selective results for their breast augmentations. The same is true for those women seeking breast reconstruction.
Dr. Turkeltaub is now also offering these breast implants for his breast augmentation and breast reconstruction patients.
About the Implants
The Natrelle INSPIRA® Cohesive breast implants can be considered to be the “gummy bear” anatomic (teardrop shaped) implants except in a round shape. They correspond exactly to the Natrelle INSPIRA® round gel-filled breast implants except are filled with an even more cohesive gel. This provides them with an even greater ability to retain their shape which can result in an improved breast appearance outcomes in certain specific situations while still preserving a soft feel. In breast augmentation, for example, some women with very thin skin may have a lower risk of visual or palpable rippling when using these implants compared to other less cohesive ones. The benefits for usage in breast reconstruction are even greater.
Cost
These highly cohesive silicone breast implants, like the gummy bear anatomic shaped ones, are priced significantly higher than the other cohesive silicone implants. This factor can impact one’s decision regarding their usage for those considering breast augmentation surgery particularly where their benefits can be considered to be negligible or nil compared to the other slightly less cohesive Natrelle INSPIRA® implants.
If you are interested in learning more about the Natrelle INSPIRA® Cohesive breast implants and how they could impact your augmentation results, schedule your consultation with Dr. Turkeltaub. Please call (480) 451-3000 or fill out our online contact form today.